
But if you are interested in exploring the 4 ‘I’s further, we discuss them in greater detail in our guide to pharmaceutical CAPA and quality management. Here are the 4 ‘I’s that should be included in any good system for CAPA in pharma in brief. A PDCA framework is tried and tested for pharmaceutical CAPA systems. 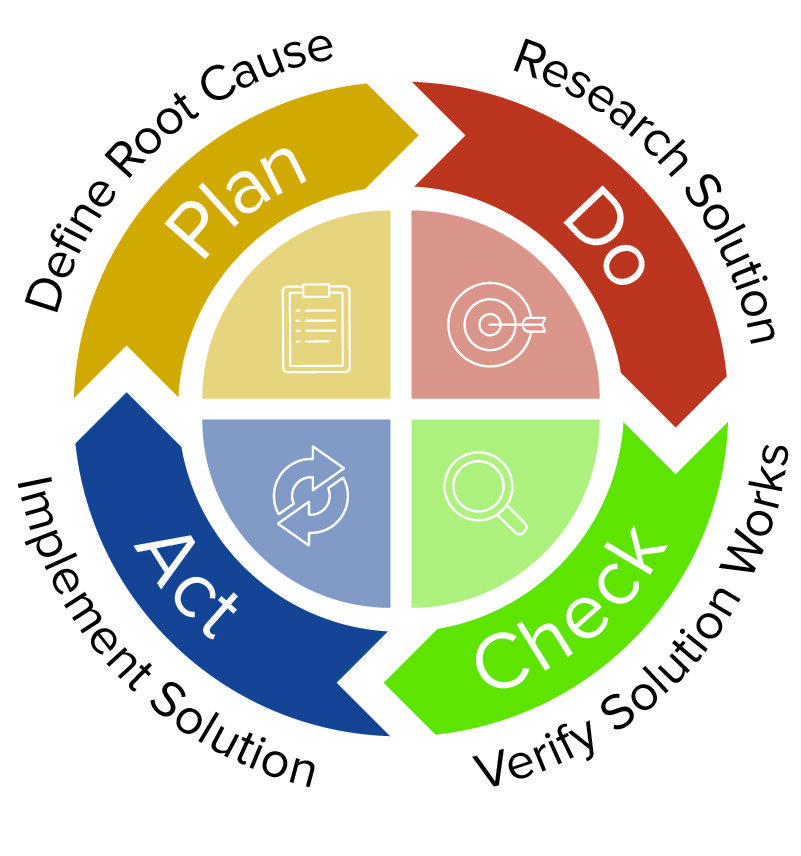 
The number of individual steps and the complexity of your documented pharmaceutical CAPA system is up to you – but several core ingredients should be in place, following a “Plan – Do – Check – Act” framework. How should you structure a pharmaceutical CAPA system? The 4 ‘I’sĪ closed-loop CAPA methodology should be implemented to deliver a standardized approach for your pharmaceutical CAPA management. Get in-depth insight into implementing effective CAPA management in pharma by reading our whitepaper Download now Want to learn more about implementing effective CAPA management? Struggling with your current pharmaceutical CAPA system? Read on for our introduction to managing CAPA in the pharmaceutical industry, or dive deeper by downloading our whitepaper on pharmaceutical CAPA below. Sub-optimal pharmaceutical CAPA management accounts for around 40% of FDA 483 submissions – you can find out more about the most common FDA 483 observations in our recent article on the subject. In short: an effective, repeatable process to put things right, and keep them right is a must-have for your quality team. Simplified adherence to quality and regulatory demands.Reduced costs thanks to reducing defects, reworks and out-of-specs.Fewer defects, reworks and out-of-specs.Improved process efficiency and patient safety.Stamping out your non-conformances and preventing them reoccurring is at the heart of continuous quality improvement, offering a range of core benefits including: Without robust control of your corrective and preventive actions (CAPAs) you open your pharmaceutical QMS, and your entire organization, up to intense scrutiny. Pharmaceutical CAPA management is a crucial competent of your quality management system (QMS).
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
